Cell and Gene Therapy Companies Leverage GenScript’s cGMP sgRNA Capability to Accelerate Speed to Market

PISCATAWAY, N.J., Dec. 15, 2022 /PRNewswire/ — GenScript USA Inc., the world’s leading life-science service provider, recently expanded its global current Good Manufacturing Practices (cGMP) single-guide RNA (sgRNA) manufacturing capabilities to meet the growing demand for cell and gene therapies. GenScript’s 400,000 square-foot cGMP facility enables next-generation gene and cell therapy R&D programs to advance IND filings and clinical trials. “We are […]
Fate Therapeutics Features Multiple Novel Approaches to Eliminate Conditioning Chemotherapy for Off-the-shelf, iPSC-derived Cell Therapies at 2022 ASH Annual Meeting

SAN DIEGO, Dec. 13, 2022 (GLOBE NEWSWIRE) — Fate Therapeutics, Inc. (NASDAQ: FATE), a clinical-stage biopharmaceutical company dedicated to the development of programmed cellular immunotherapies for patients with cancer, presented preclinical data of several novel strategies designed to enable administration of off-the-shelf cell-based cancer immunotherapies without conditioning chemotherapy at the 64th American Society of Hematology […]
GT Biopharma and Fate Therapeutics Present Preclinical Data Highlighting Novel Dual Antigen Targeting Approach For The Treatment of AML at ASH 2022

BRISBANE, CALIFORNIA, Dec. 12, 2022 (GLOBE NEWSWIRE) — GT Biopharma, Inc. (NASDAQ: GTBP) today announced the presentation of new preclinical data at the American Society of Hematology’s 64th Annual Meeting (ASH 2022). The presentation highlights the potential of a novel dual antigen targeting approach for the treatment of acute myeloid leukemia (AML) by combining GT […]
IN8bio Announces FDA Clearance to Initiate a Phase 2 Clinical Trial of INB-400 Gamma-Delta T Cells for Glioblastoma

NEW YORK, Dec. 08, 2022 (GLOBE NEWSWIRE) — IN8bio, Inc. (Nasdaq: INAB), a clinical-stage biopharmaceutical company developing innovative gamma-delta T cell therapies, today announced that it has received clearance of its Investigational New Drug (IND) application from the U.S. Food and Drug Administration (FDA) to initiate a Phase 2 clinical trial of a genetically modified […]
Leading Stem Cell Clinic in the US Develops First Real “Stem Cell Face Mask”
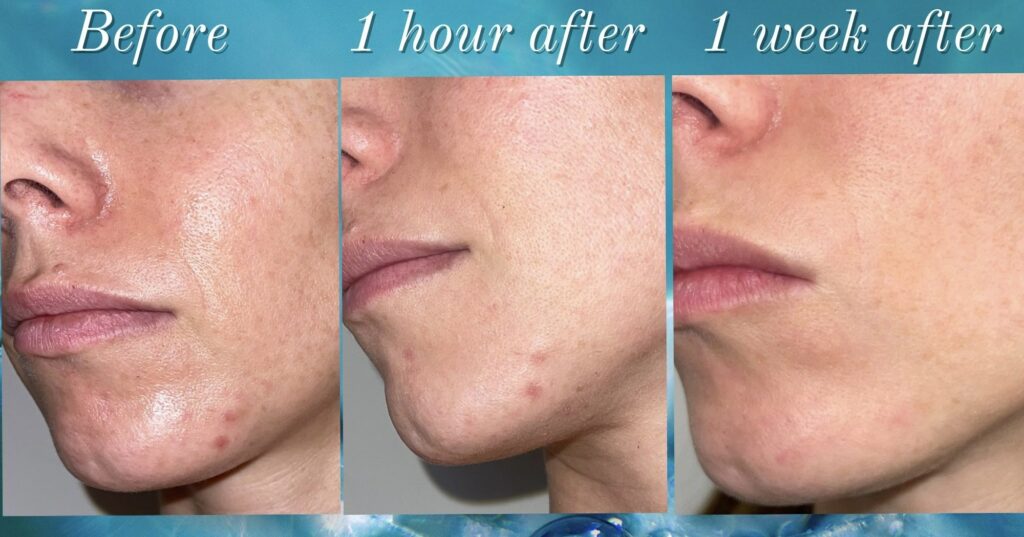
LOS ANGELES, Dec. 6, 2022 /PRNewswire/ — Springs Rejuvenation began as a Stem Cell and Exosome treatment center with a focus on acute medical conditions and joint injures. More recently the springs team began focusing on skin health adding exosome therapy to already popular treatments such as “The Vampire”/PRP facial, micro needling and non surgical hair transplant. With the […]
BioRestorative Therapies Announces SBIR Phase I Grant to Explore the Therapeutic Effects of ThermoStem® Targeting Polycystic Ovary Syndrome as an Indication

MELVILLE, N.Y., Dec. 06, 2022 (GLOBE NEWSWIRE) — BioRestorative Therapies, Inc. (“BioRestorative”, “BRTX” or the “Company”) (NASDAQ:BRTX), a clinical stage company focused on stem cell-based therapies, today announced that it has been awarded a Small Business Innovation Research (SBIR) Phase I grant from the Eunice Kennedy Shriver National Institute Of Child Health & Human Development of the […]
Lyell Immunopharma to Participate in BofA Securities Biotech Conference

SOUTH SAN FRANCISCO, Calif., Dec. 01, 2022 (GLOBE NEWSWIRE) — Lyell Immunopharma, Inc. (Nasdaq: LYEL), a clinical‑stage T-cell reprogramming company dedicated to developing curative cell therapies for patients with solid tumors, today announced that members of its senior management team will participate in the virtual BofA Securities 2022 Biotech SMID Cap Conference on Thursday, Dec. 8 […]
2022 Ogawa-Yamanaka Stem Cell Prize Awarded to Juan Carlos Izpisua Belmonte – Award Ceremony

On November 17, 2022, Gladstone Institutes honored the 2022 Ogawa-Yamanaka Stem Cell Prize winner, Juan Carlos Izpisua Belmonte. Izpisua Belmonte is a celebrated scientist known for his regenerative medicine and aging research, as well as his work leading to improved methods of generating induced pluripotent stem cells (iPSCs). In a translational leap, his group was […]
reNEW | Next generation stem cell research

reNEW is a unique global collaboration focusing on stem cell research involving three leading research institutions: the University of Copenhagen in Denmark, the Leiden University Medical Center in the Netherlands, and the Murdoch Children’s Research Institute in Australia. reNEW’s vision is to create a new generation of effective, safe, and socially sustainable stem cell-based therapies […]
Merakris Announces Review in Biomedicines Medical Journal Highlighting Therapeutic Potential of Cell-Free Amniotic Fluid

RESEARCH TRIANGLE PARK, N.C., Nov. 29, 2022 (GLOBE NEWSWIRE) — A review published recently in the international scientific journal Biomedicines highlights the therapeutic benefits of amniotic fluid in restoring, repairing and regenerating damaged tissue across a range of human diseases and disorders. The article – Cell-Free Amniotic Fluid and Regenerative Medicine: Current Applications and Future Opportunities https://www.mdpi.com/2227-9059/10/11/2960 – evaluates a […]